
In such cases, chemists usually define a standard by arbitrarily assigning a numerical value to one of the quantities, which allows them to calculate numerical values for the rest.įigure 1.6.2 Determining Relative Atomic Masses Using a Mass Spectrometer. We will encounter many other examples later in this text. It is actually rather common in chemistry to encounter a quantity whose magnitude can be measured only relative to some other quantity, rather than absolutely. Thus it is not possible to calculate absolute atomic masses accurately by simply adding together the masses of the electrons, the protons, and the neutrons, and absolute atomic masses cannot be measured, but relative masses can be measured very accurately. By measuring the relative deflection of ions that have the same charge, scientists can determine their relative masses (Figure 1.6.2). The extent of the deflection depends on the mass-to-charge ratio of the ion. When an electric field is applied, the ions are accelerated into a separate chamber where they are deflected from their initial trajectory by a magnetic field, like the electrons in Thomson’s experiment. First, electrons are removed from or added to atoms or molecules, thus producing charged particles called ions. The technique is conceptually similar to the one Thomson used to determine the mass-to-charge ratio of the electron. The relative atomic mass is an average of the atomic masses of all the different isotopes in a sample, with each isotopes contribution to the average determined by how big a fraction of the sample it makes up. The longest-lived radioisotope is 32 Si, which is produced by cosmic ray spallation of argon. 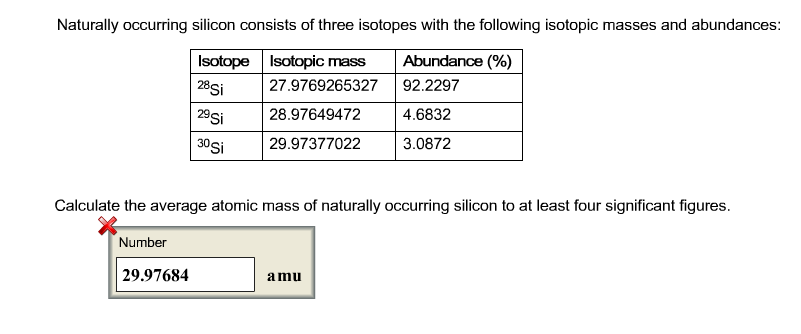
Atomic mass of Silicon (Si) 28.085: 28: 15: Atomic mass of. Given that the average atomic mass of silicon is 28. The most abundant isotope is 28Si, which accounts for 92.23 of naturally occurring silicon. Scientists can measure relative atomic masses very accurately, however, using an instrument called a mass spectrometer. Silicon ( 14 Si) has 23 known isotopes, with mass numbers ranging from 22 to 44. Atomic mass of all elements (along with the rounded off values) is mentioned in the chart below. Naturally occurring silicon exists as 3 isotopes: 28Si, 29Si, and 30Si, whose atomic masses are 27.9769, 28.9865, and 29.9838, respectively. We can easily calculate the binding energy from the mass difference using Einstein's formula E=mc 2.īecause atoms are much too small to measure individually and do not have a charge, there is no convenient way to accurately measure absolute atomic masses. The silicon molar mass of NBS-28 is determined to be 28.08653(11), which is 0.001 larger than previously reported values. Although the difference in mass is small, it is extremely important because it is the binding energy of the nucleus. For example, the ratio of the masses of 1H (hydrogen) and 2H (deuterium) is actually 0.500384, rather than 0.49979 as predicted from the numbers of neutrons and protons present. The atomic mass of an element is the average mass of the atoms of an element measured in atomic mass unit. X Research source This means Ag-109 has two more neutrons per atom than Ag-107, giving it slightly more mass.Br\) or, more commonly, 79Br and 81Br.Īlthough the masses of the electron, the proton, and the neutron are known to a high degree of precision (Table 1.5.1), the mass of any given atom is not simply the sum of the masses of its electrons, protons, and neutrons. X Research source Isotopes are named after the "mass number," or the sum of protons and neutrons in one atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. For example, the element silver (Ag) has two naturally occurring isotopes: Ag-107 and Ag-109 (or 107Ag and 109Ag).The average atomic mass of the element takes the variations of the number of neutrons into account, and tells you the average mass per atom in a typical sample of that element.X Trustworthy Source Science Direct Online archive of peer-reviewed research on scientific, technical and medical topics Go to source However, the element always has the same number of protons. X Research source The only difference between two isotopes of the same element is the number of neutrons per atom, which affects the atom's mass. Each proton and each neutron weigh 1 atomic mass unit (amu). The mass number for each isotope is the sum of numbers of protons and neutrons in the nucleus. Most elements can naturally occur in multiple forms, or isotopes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
